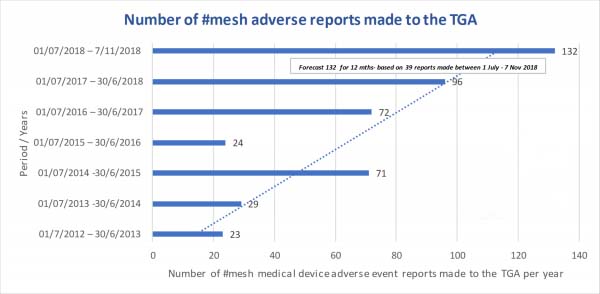
Chart above is based on TGA “mesh” search on IRIS database of medical device incident reports. Please note that the current financial year is forecast based on the numbers reported between July and Nov 2018. We ARE making an impact. Each and every one of YOU.
Why is it important to report your injury to the TGA?
At the time of writing this piece, it is NOT mandatory for your surgeon or GP to report your mesh injury to the TGA.
Without the availability of statistics in this area, it is difficult to lobby for assistance. No one knows how many (possibly tens of thousands) of mesh injured there are in Australia. Many are likely to be over the age of 55, not on social media, and without any kind of “mesh implant register”, receive no follow up or knowledge that their ongoing health issues may be attributable to mesh implanted for prolapse, stress urinary incontinence, or hernia.
That’s where you come in!
Your “statistic” matters. In fact, we need you to report your “adverse event” to the TGA, to add to the statistics that are kept and reported to Government agencies.
It looks daunting, but it isn’t. If you have your records from your mesh implant, most information will be there. Or you can reach out to a FB support group and ask someone who has submitted theirs to lend a hand.
Thank you to all of those who have already made this very important step. It is truly appreciated.
Frequently Asked Questions
Patient’s Guide to Reporting
The form is too long and complicated.
Volunteers will can help you work through it. Generally, when assisted, it takes around 30 minutes!!
I don’t have a computer.
Well we do! You can relay the information to our volunteer, and still submit your form.
I don’t know what they mean by ‘supplier’ on the form.
Our volunteers have recorded all the suppliers of past and present available mesh products in Australia, and their addresses.
I don’t know what type of implant I have.
You can still report your complication based on the information that you do have. We can help narrow down the possibilities and assist in filling the form out as best possible.
I don’t want my doctor to find out.
You do not give the name of the implanting surgeon or your doctor on the form. So they don’t even have to know.
I’m really not that bad.
The TGA is required to document all adverse events, whether it be pain, foreign body response, surgical injury, infection, erosion, or even persistent discomfort. They are even obliged to document any near miss!
I don’t want to cause trouble.
Reporting an adverse event is not causing trouble, it is following correct process. That is what it is set up for.
I don’t understand the abbreviations. I don’t even know what TGA is.
The TGA stands for Therapeutic Goods Administration. It is THE regulatory body that allows medicines and implants to be used in Australia. It is there job to engage in surveillance of all health products.
I think my doctor should do that.
Good point, but most don’t. That is why the TGA permits direct consumer reporting.
TGA list of complications possibly associated with mesh taken directly from the TGA website. Adverse events that may be associated with urogynaecological meshes include:
- punctures or lacerations of vessels, nerves, structures or organs, including the bladder, urethra or bowel (these may require surgical repair)
- transitory local irritation at the wound site
- a ‘foreign body response’ (wound breakdown, extrusion, erosion, exposure, fistula formation and/or inflammation)
- mesh extrusion, exposure, or erosion into the vagina or other structures or organs
- as with all foreign bodies, mesh may potentiate an existing infection
- over-correction (too much tension applied to the tape) may cause temporary or permanent lower urinary tract obstruction
- acute and/or chronic pain
- voiding dysfunction
- pain during intercourse
- neuromuscular problems including acute and/or chronic pain in the groin, thigh, leg, pelvic and/or abdominal area
- recurrence of incontinence
- bleeding including haemorrhage, or haematoma
- seroma
- urge incontinence
- urinary frequency
- urinary retention
- adhesion formation
- atypical vaginal discharge
- exposed mesh may cause pain or discomfort to the patient’s partner during intercourse
- mesh migration
- allergic reaction
- abscess
- swelling around the wound site
- recurrent prolapse
- contracture
- scarring
- excessive contraction or shrinkage of the tissue surrounding the mesh
- vaginal scarring, tightening and/or shortening
- constipation/defecation dysfunction
- granulation tissue formation